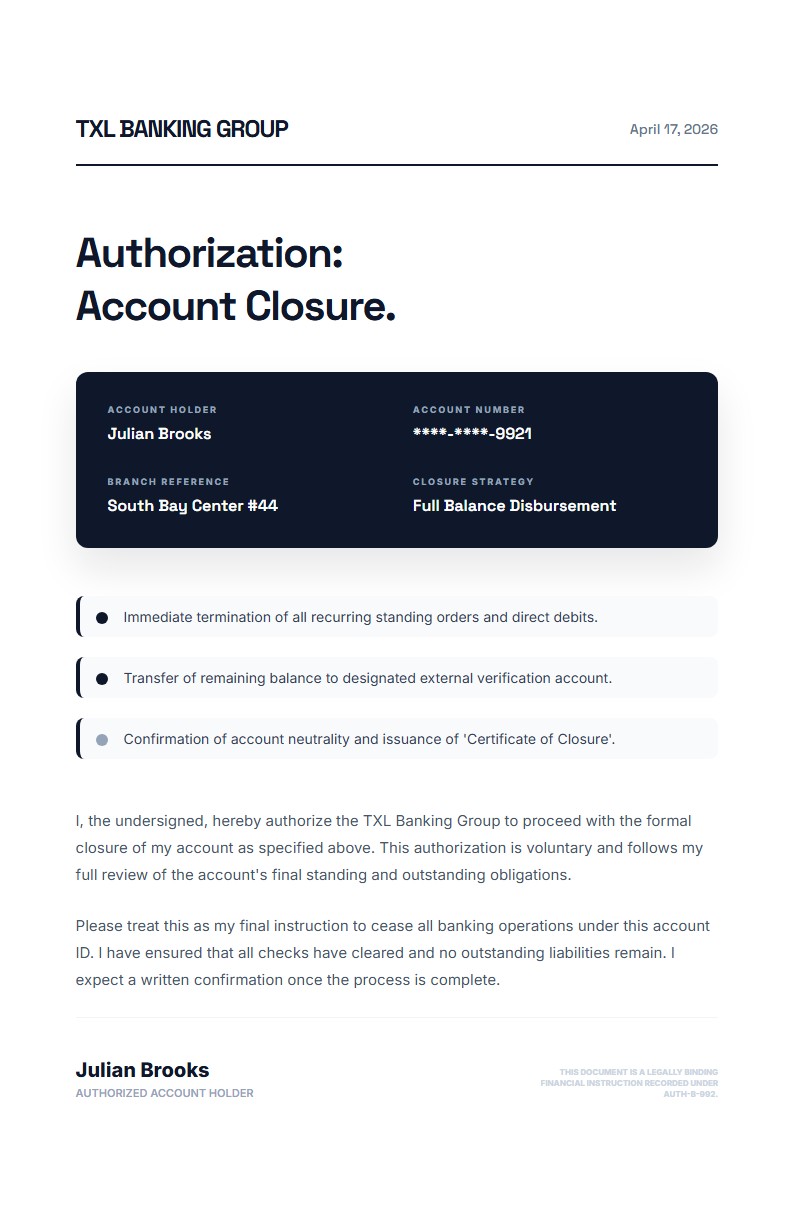
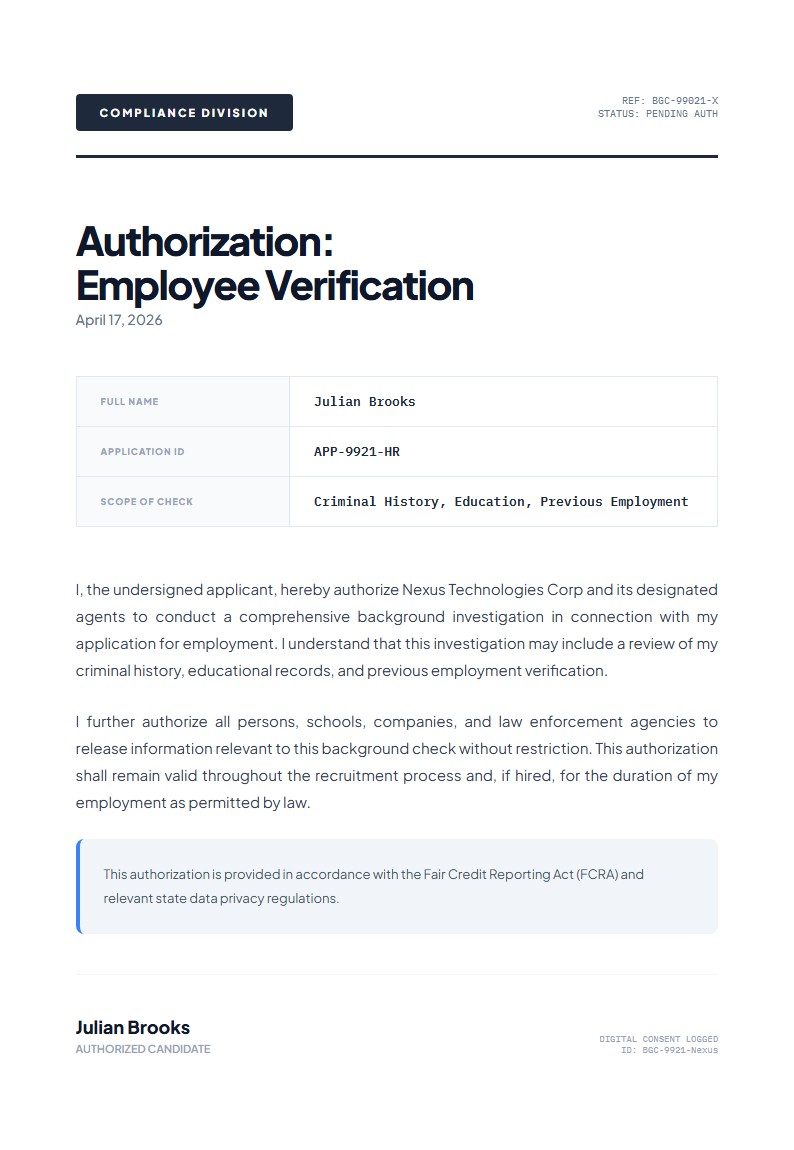
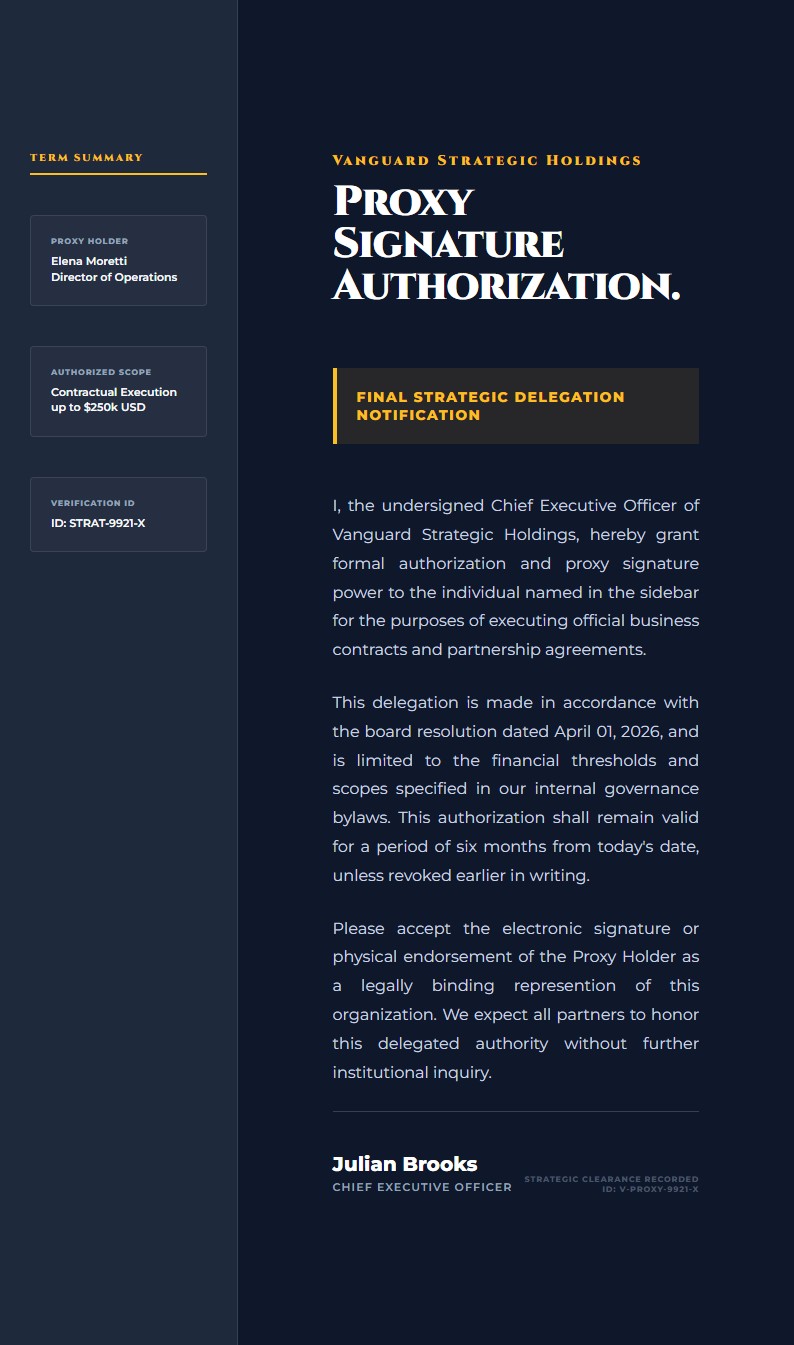
Clinical Trial Participation Authorization
Use this when you are a legal guardian or parent authorizing a minor's participation in a clinical research study. It is designed to be sensitive and scientific, providing the 'Protocol ID' and ethical 'IRB' context required by research institutions and medical boards.
A sensitive medical design featuring a 'Soft Crimson' palette, 'Plus Jakarta Sans' and 'IBM Plex Mono' typography, and a series of delicate 'Pulse Wave' SVG motifs. The layout prioritizes ethical oversight with a specialized 'Participant Card' and includes a strike 'Ethical Callout' box, creating an atmosphere of professional intellectual rigor and deep empathetic care.
Katalisx helps you manage health outcomes with precision and heart. This template ensures that your clinical trial authorization is presented as a methodical scientific permit, providing all necessary protocol identification and informed consent references needed for immediate compliance with medical ethics committees and research boards.
A guardian authorization for a child participating in a pediatric clinical trial with formal consent documentation.
Use for guardian consent when a teenager is being enrolled in a structured clinical or medical research study.
A careful authorization letter for a guardian allowing minor participation in a monitored clinical trial enrollment process.

Choose the perfect template from our professional library that fits your specific needs.
Fill in the details. Changes are reflected instantly in the live letter preview.
Once satisfied, download your professional-grade PDF, ready for immediate use.
Hand-picked Authorization Letter alternatives for your professional needs.