Academic/Research Data Access Authorization
Use this when a principal investigator, research director, or institutional authority needs to authorize controlled access to data for academic or scientific work. It works well for clinical datasets, restricted research archives, and sensitive project repositories where ethical oversight and formal access tiers must be clearly documented.
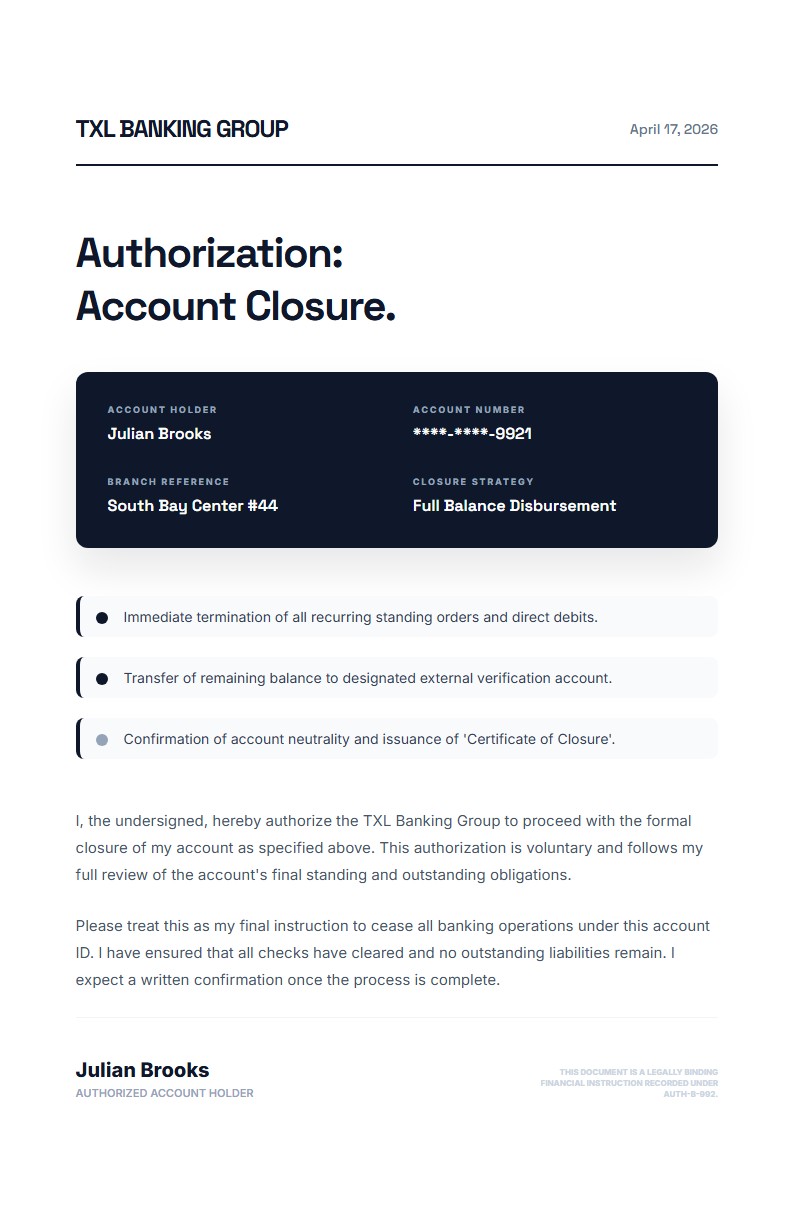
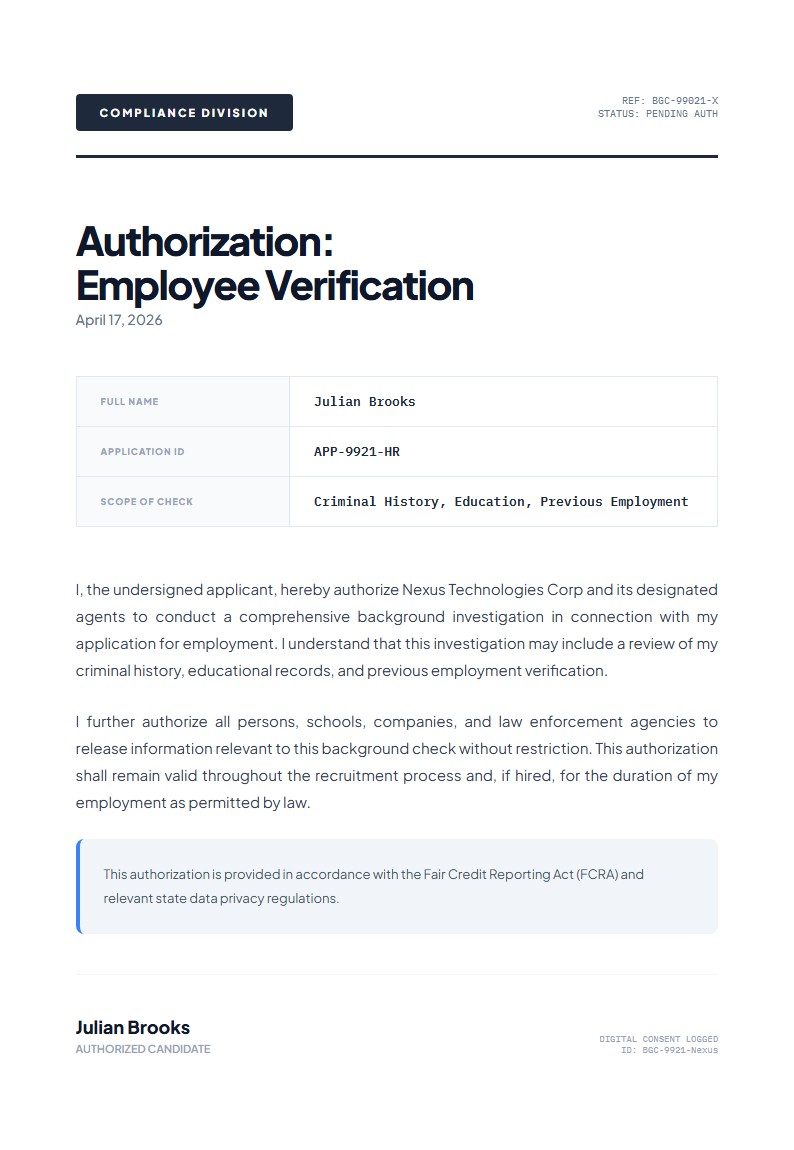
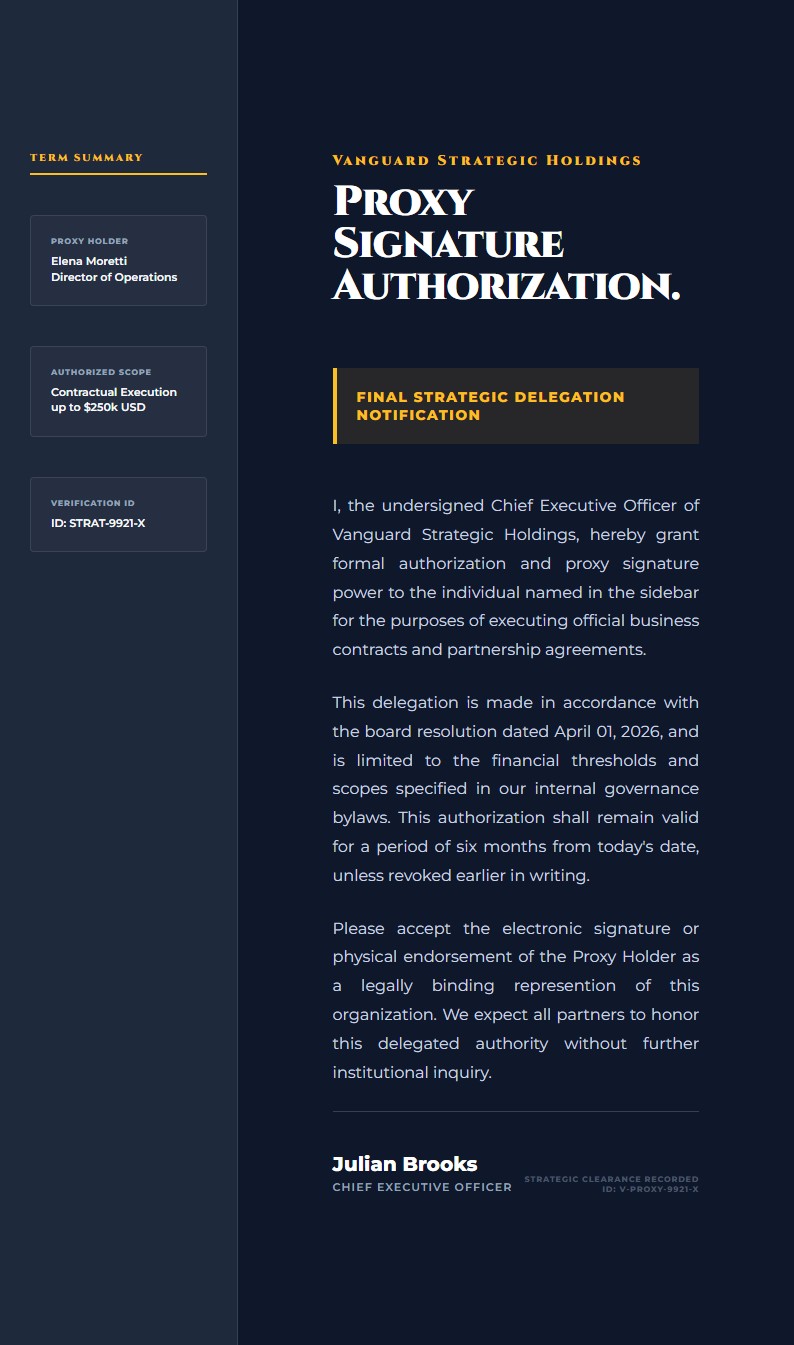
The layout is formal and compliance-focused, with a research access grid that highlights the principal investigator, protocol reference, and access tier alongside a separate security notice. That makes it especially useful for authorization letters that need to balance permission with clear governance conditions.
Katalisx helps you present a research data access authorization in a way that feels rigorous, compliant, and institution-ready. Instead of a loose permission note, you get a format that clearly states who is authorized, under what protocol, and with what conditions.
Authorize access to anonymized clinical or patient-linked research data.
Authorize a researcher to access a restricted archive or protected study repository.
Authorize access to a shared data repository for a multi-institution research project.

Choose the perfect template from our professional library that fits your specific needs.
Fill in the details. Changes are reflected instantly in the live letter preview.
Once satisfied, download your professional-grade PDF, ready for immediate use.
Hand-picked Authorization Letter alternatives for your professional needs.